Quality Cloud
Integrated Quality Management Software
- Automate Quality Processes. Digitize audits, inspections, and workflows to reduce errors and save time.
- Ensure Product Compliance. Maintain audit readiness and meet global standards with centralized oversight.
- Strengthen Supplier Performance. Monitor suppliers, track risks, and drive accountability across the value chain.
Trusted for performance advantage worldwide
All Quality Software Solutions & Related Capabilities
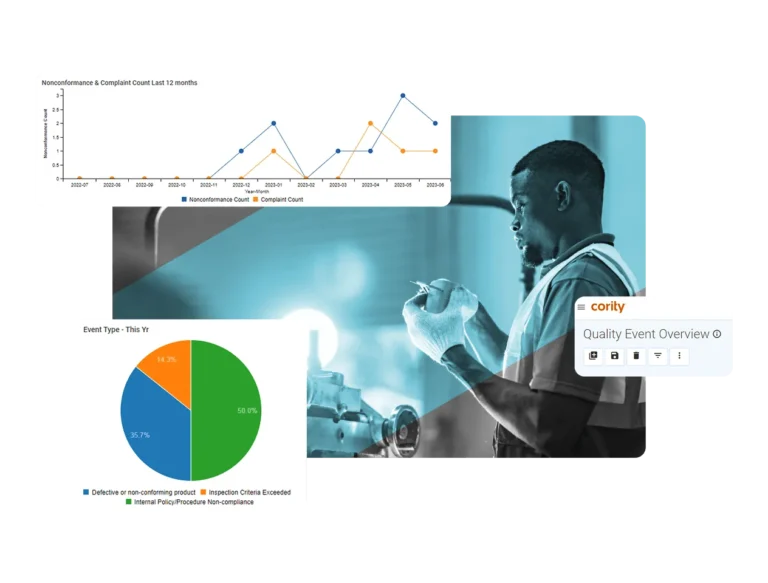
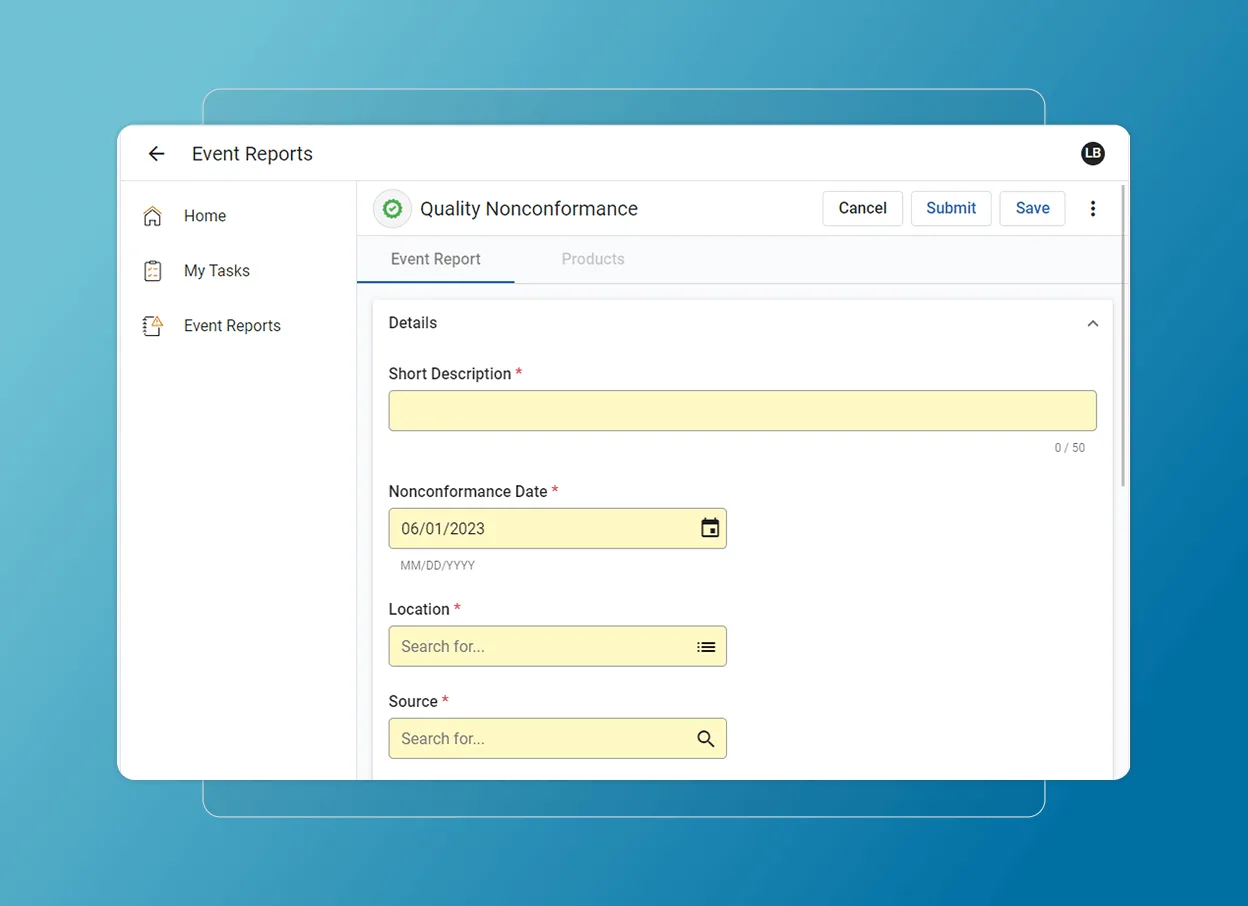
Capture and manage issues
Track and resolve nonconformances from inspections, audits, and supplier data with a closed-loop system that improves quality management.
Automate workflows
Save time by automating the nonconformance lifecycle with pre-configured workflows and notifications.

Improve traceability
Close gaps quickly




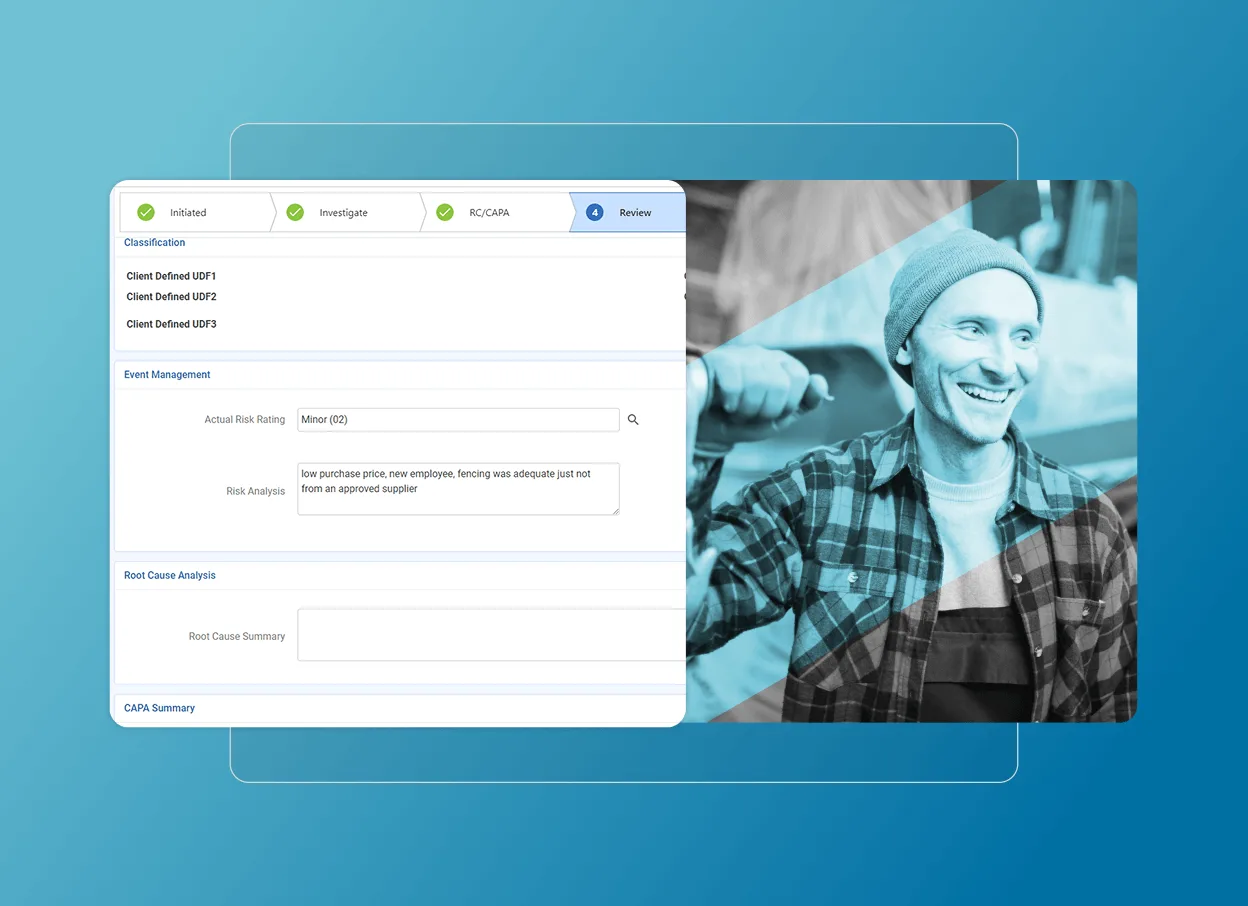
Streamline corrective actions
Manage and assign internal, external, and supplier corrective actions with an integrated solution designed for accuracy and compliance.

Improve accountability
Automate documentation
Prevent repeat issues
Identify root causes and link to specific actions, ensuring the long-term resolution of quality management problems.





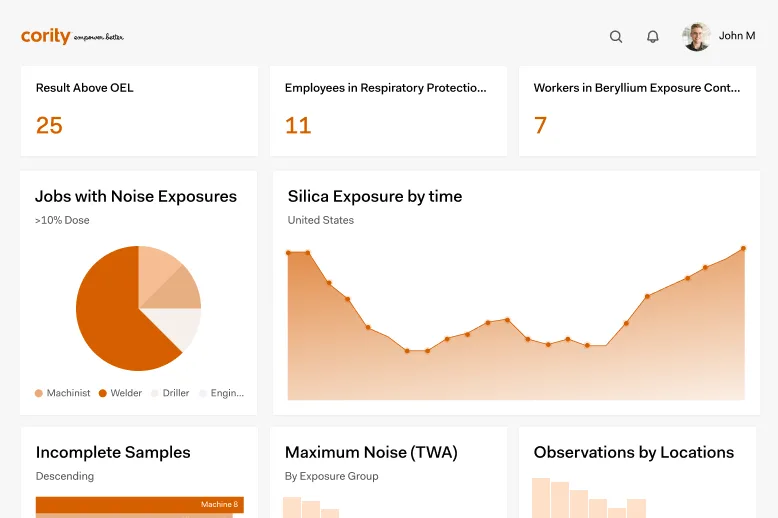
Track supplier management performance
Improve supply chain quality
Automate supplier approvals workflows
Predict and prevent risk




Centralize documentation
Store all quality-related documents securely in a single system for easy access and version control.

Automate version control
Improve document access
Simplify audits


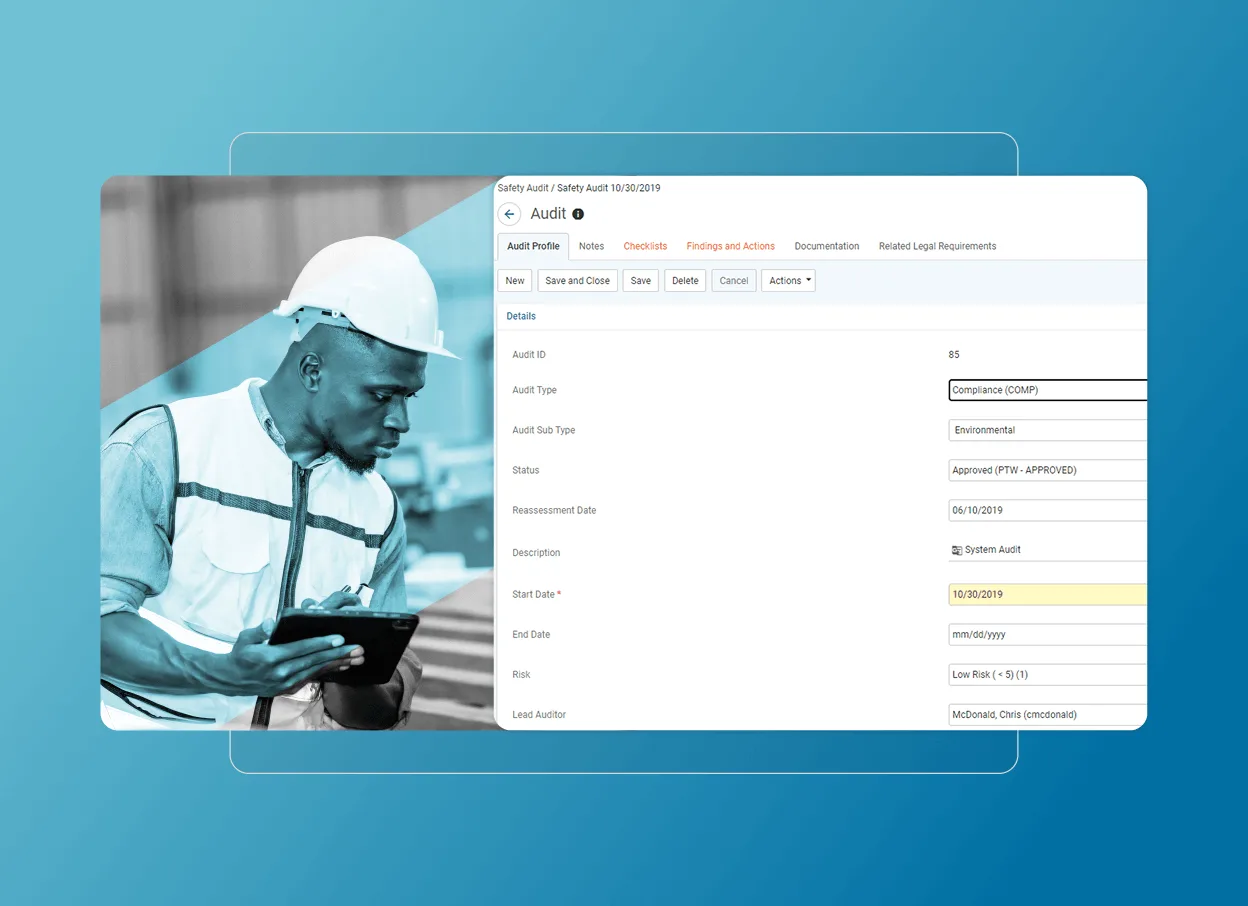
Capture data on the go
Real-time syncing
Improve engagement
Streamline inspections



4.3
Award Winning Quality Management Solutions
Recognized globally for innovation and operational excellence




How it Works
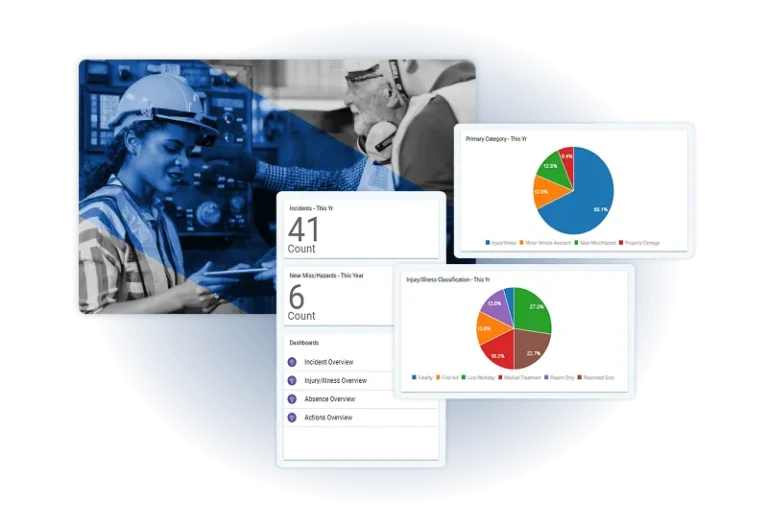
Step 1: Centralize Quality Data
Unify product, supplier, and audit data in one platform for complete visibility, faster decision-making, and stronger compliance readiness.

Step 2: Automate Workflows
Streamline audits, inspections, nonconformance management, and CAPAs with configurable workflows that reduce manual effort and close gaps faster.
Step 3: Continuously Improve
Leverage dashboards and analytics to identify trends, strengthen supplier performance, and reduce the overall cost of quality management.
Trusted globally for quality excellence
85%
ISO audit preparation reduced from 2 weeks to 2 hours
40%
Faster supplier response times by a precision machining leader
28%
Cost of Poor Quality reduced in 1 year by Outdoor Power Equipment Manufacturer
Leading Quality Management Software Solutions
Nonconformance Management
Capture, track, and resolve product issues with quality management software designed for speed and traceability.
Corrective & Preventive Actions
Improve accountability and prevent recurring issues with integrated quality software and automated CAPA workflows.
Supplier Quality Management
Enhance supply chain collaboration, reduce risk, and improve outcomes with connected quality management solutions.

Cority Launches Rapidly Deployable Quality Essentials Solution to Enhance Cross-Functional EHS Management
Software helps organizations quickly reduce costs, ensure compliance, and safeguard reputation Toronto, ON, June 22, 2023 – Leading global EHS

Cority Announces QMS Partnership with GEODIS
Global logistics leader selects software provider’s Quality Management Solution to manage customer complaints and improve customer satisfaction Toronto, ON, October

Cority Expands Quality Management Solution to Support the Product Development Process
New functionality streamlines product quality and supplier collaboration to reduce nonconformances and drive continuous improvement efforts Toronto, Canada – November
Real Stories. Smarter EHS+ Insights.
Nicholas Sondelski
Information Systems Administrator
Before, our response time for suppliers was about 5 days. We’ve seen their response time drop down to around 3 days.
Doug Jones
Quality Assurance Manager
“We use quality as a weapon. The data is always there, ins available at our fingertips to support our position.”
Pierre Perrier
National Manager of Transportation Safety
“Cority has allowed us to identify our problems and build programs to help drive prevention in those areas.”

Unlock the Power of Quality Management Software
Elevate product quality, supplier collaboration, and compliance with modern quality software solutions. Automate workflows, reduce costs, and continuously improve performance with a single connected platform built for lasting results.





